









Copyright 2009-2014 Mahavir Gems. All Rights Reserved.
MAHAVIR GEMS
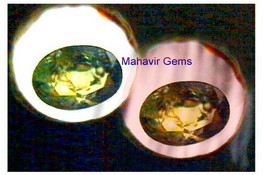
Alexandrite
Chrysoberyl .
Among gem minerals chrysoberyl ranks third in the list of hardness (H 8½), next only to diamond (H 10) and corundum (H 9). Alexendrite that changes colour in different light source, Cat's eye or cymophane (in Greek meaning appearing like a wave, in Sanskrit it is known as Vaidurya) and common transparent greenish yellow type are the gem varieties of chrysoberyl.
Astrologically Hindus regard Vaidurya as the stone for Ketu or dragon's tail. The fascinating alexandrite, a mineral chameleon, has gained some importance in India only in recent years, although it has been a highly esteemed stone in western countries for more than a century.
Commonly occurring yellowish or greenish coloured transparent chrysoberyl, however, is not favoured much in the trade. With the discovery of new gem deposits in Orissa, Andhra Pradesh, Madhya Pradesh and Kerala, chrysoberyl has attained considerable importance in India.
Alexandrite - Important Characteristics
- Classification
- Hardness (Mohs Scale)
- Chem. formula
- Composition
- Crystal Structure
- Colour
- Atomic (Crystal )Structure
- Index of Refraction
- Density (Relative)
- Luster
- Uses
: Mineral
: Eight & half (8.5 )
: BeAl2O4
: Beryllium Aluminium Oxide.
: Intergrown Triplets.
: All Shades of Green.
: Orthorhombic
: 1.746 - 1.763
: 3.70 - 3.78
: Vitreous
: Jewelry, decorative art, ornaments.
Chemical composition of chrysoberyl is BeAI2O4. Iron impurity is usually present in traces resulting in shades of yellow, brown and green. However, the presence of chromium dictates a dramatic change in colour. The other properties are, SG = 3.71; lustre - vitreous; RI, a = 1.744-1.74, ß = 1.747-1.749, ? = 1.753-1.758; d = 0.009-0.011; optic sign B+; pleochroism = weak (in ordinary variety) to strong (in alexandrite). Fluid inclusions and liquid feathers are often encountered.
Varieties of Chrysoberyl
Three gem varieties can be recognised namely common transparent chrysoberyl, alexandrite and cymophane.
Alexandrite:-This gem variety is known for dichromatism/metamerism being distinctly green in daylight or fluorescent lamp and reddish or purplish in incandescent light. It was first recognized in 1830 at the emerald mines near Takovaya, UraI Mts and was named after Alexander II, the Russian Czar. In some alexandrite, colour changes from green in daylight to greyish or brownish in incandescent light. The colour and metamerism is derived from Cr3+ that replaces Al3+ in octahedral site. Under Chelsea filter and crossed filters it appears red. Even opaque specimens having colour change from green to grey from Orissa and Kerala also appear reddish through these filters.
Alexandrite is strongly pleochroic; X = red to purple, Y = yellow to orange and Z = green. Spectroscopic examination reveals distinct doublet in the deep red (680.3 and 678.5nm) and a few other narrower lines. Only those chrysoberyls that exhibit doublet in deep red are considered as alexandrite. Liquid feather inclusions can be seen in some alexandrites.
Simulants of alexandrite include natural and synthetic vanadium corundum and spinel. Since many of these vanadium bearing stones do contain chromium, they appear red under Chelsea filter. Colour change in these stones is generally from greenish or brownish grey or bluish violet in daylight to amethystine or purple-red in incandescent light. While trichroism is shown by alexandrite. vanadium corundum exhibits dichroism. Alexandrite is produced by flux-fusion and floating zone methods and Czochralski 'pulling' technique. These synthetics display startling colour change from green to raspberry red. While most natural alexandrites are usually highly flawed with fracture and inclusions, the synthetics are barely flawed. They may consist of liquid feathers and curved striae. Genuine (natural) alexandrite may also reveal liquid feather like inclusions.
Alexandrite
(Left in Daylight & Right in Artifical light)
This site is best viewed on 1024 x 768 screen resolution using Mozilla 3.5 Browser


Alexandrite
(Rough Crystal of a Alexandrite)
Alexandrite is named for the former czar of Russia, Alexander II, and was first discovered in the Ural Mountains of Russia, supposedly on the day of his birth.
Chromium gives alexandrite its color and while in most minerals a trace element like chromium would provide only one color to the mineral, in alexandrite it gives it two! Coloring agents are dependent on the wavelength of light and the chemical bonds in the crystal to determine the color that they will cause.
An element like copper, in normal light, can cause a green color in malachite and a blue color in azurite, it all depends on the character of the chemical bonding.
In a single specimen of alexandrite, the chromium is in such a balanced situation that the color of the specimen depends on the character of light that hits the crystal. If the light is natural sunlight or fluorescent light, the crystal will be green; however, if the light is incandescent light from a common indoor light bulb, then the crystal will appear red.
Beaware of Synthetic corundums spiked with trace elements that yield an alexandrite-like color change are sold as alexandrite in the gemstone markets. Many people fall pray to such malpractice.
Always demand a Certified Gemstones whereby the certificate is issued by a Reputed Laboratory or a Reputed Gemologist
These stones have a red-violet, near-amethyst color in incandescent light and a blue-violet color in daylight. They are far cheaper than natural alexandrites, which are some of the rarest and most expensive of gemstones.
Alexandrite is the modern June birthstone.



